|
There are a variety of radioactive decay modes. Each decay event involves loss of mass or charge. This event is called a nuclear transformation, a decay, or a disintegration. Brookhaven National Laboratory.Radioactivity is the spontaneous emission of charged particles or photons by an atomic nucleus that is in an unstable configuration. Half-life, spin, and isomer data selected from the following sources.International Union of Pure and Applied Chemistry. "News & Notices: Standard Atomic Weights Revised"."Atomic weights of the elements 2005 (IUPAC Technical Report)". de Laeter, John Robert Böhlke, John Karl De Bièvre, Paul Hidaka, Hiroshi Peiser, H.Isotopic compositions and standard atomic masses from:.Audi, Georges Bersillon, Olivier Blachot, Jean Wapstra, Aaldert Hendrik (2003), "The N UBASE evaluation of nuclear and decay properties", Nuclear Physics A, 729: 3–128, Bibcode: 2003NuPhA.729.3A, doi: 10.1016/j.nuclphysa.2003.11.001."Five-year cure of cervical cancer treated using californium-252 neutron brachytherapy". ^ "Californium-252 & Antimony-Beryllium Sources"."Production, distribution and applications of californium-252 neutron sources". ^ "Portable Isotopic Neutron Spectroscopy (PINS) for the Military".^ Excluding those " classically stable" nuclides with half-lives significantly in excess of 232Th e.g., while 113mCd has a half-life of only fourteen years, that of 113Cd is nearly eight quadrillion years.^ This is the heaviest nuclide with a half-life of at least four years before the " sea of instability".No alpha activity attributable to the new isomer has been detected the alpha half-life is probably greater than 300. No growth of Cf 248 was detected, and a lower limit for the β − half-life can be set at about 10 4. This was ascribed to an isomer of Bk 248 with a half-life greater than 9. "The isotopic analyses disclosed a species of mass 248 in constant abundance in three samples analysed over a period of about 10 months. "The alpha half-life of berkelium-247 a new long-lived isomer of berkelium-248". ^ Specifically from thermal neutron fission of uranium-235, e.g.Radium's longest lived isotope, at 1,600 years, thus merits the element's inclusion here. While actually a sub-actinide, it immediately precedes actinium (89) and follows a three-element gap of instability after polonium (84) where no nuclides have half-lives of at least four years (the longest-lived nuclide in the gap is radon-222 with a half life of less than four days). Chinese Physics C, High Energy Physics and Nuclear Physics. "The NUBASE2020 evaluation of nuclear physics properties *". "α decay of Fm 243 143 and Fm 245 145, and of their daughter nuclei". "The new isotope 236Cm and new data on 233Cm and 237, 238, 240Cf" (PDF). National Nuclear Data Center, Brookhaven National Laboratory. sfn error: no target: CITEREFCRC2006 ( help) In patients with certain types of brain and cervical cancer, 252Cf can be used as a more cost-effective substitute for radium. Medicine Ĭalifornium-252 has also been used in the treatment of serious forms of cancer. Instrumentation is lowered into the well which bombards the formation with high energy neutrons to determine porosity, permeability, and hydrocarbon presence along the length of the borehole. 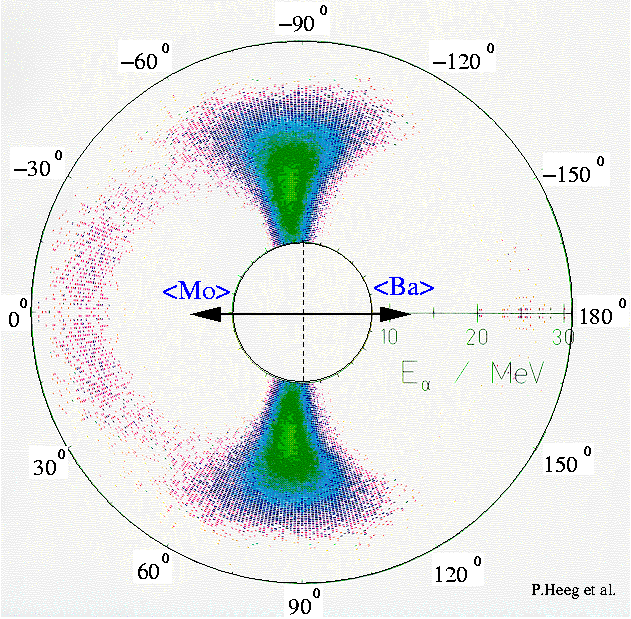
In the oil industry, 252Cf neutron sources are used to find layers of petroleum and water in a well. Customs and Border Protection, employs the use of 252Cf sources to detect hazardous contents found inside artillery projectiles, mortar projectiles, rockets, bombs, land mines, and improvised explosive devices (IED). 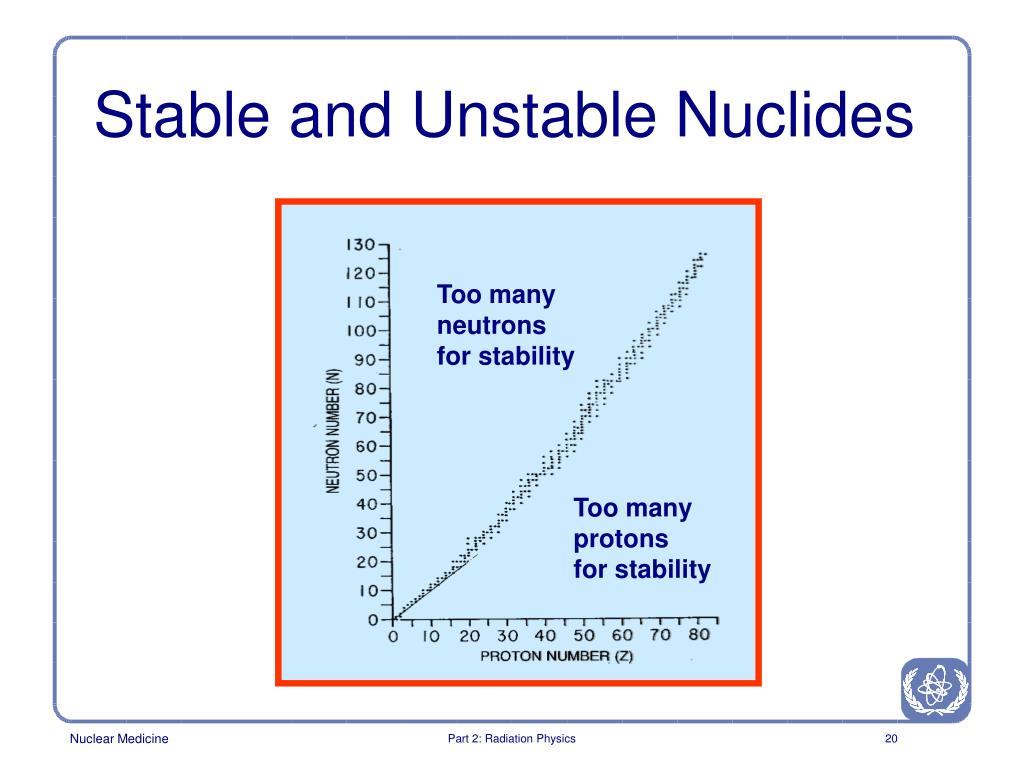
The portable isotopic neutron spectroscopy (PINS) used by United States Armed Forces, the National Guard, Homeland Security, and U.S. Once a reactor is filled with nuclear fuel, the stable neutron emissions from the source material initiates the fission chain reaction. The neutron sources produced from 252Cf are most notably used in the start-up of nuclear reactors. 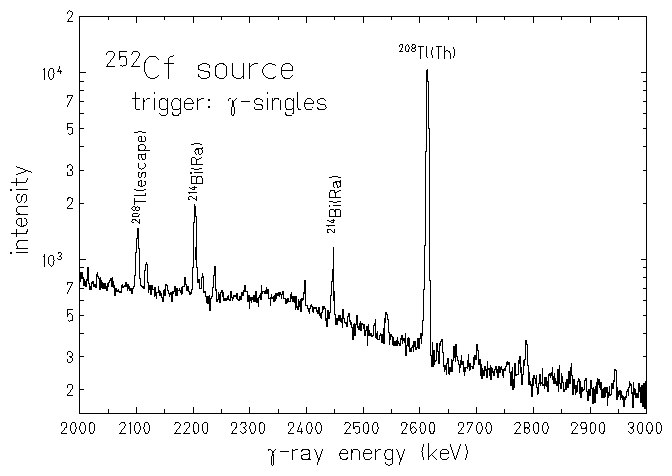
This isotope produces high neutron emissions and can be used for a number of applications in industries such as nuclear energy, medicine, and petrochemical exploration. Fission neutrons have an energy range of 0 to 13 MeV with a mean value of 2.3 MeV and a most probable value of 1 MeV. Californium-252 (Cf-252, 252Cf) undergoes spontaneous fission with a branching ratio of 3.09% and is used in small sized neutron sources.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
